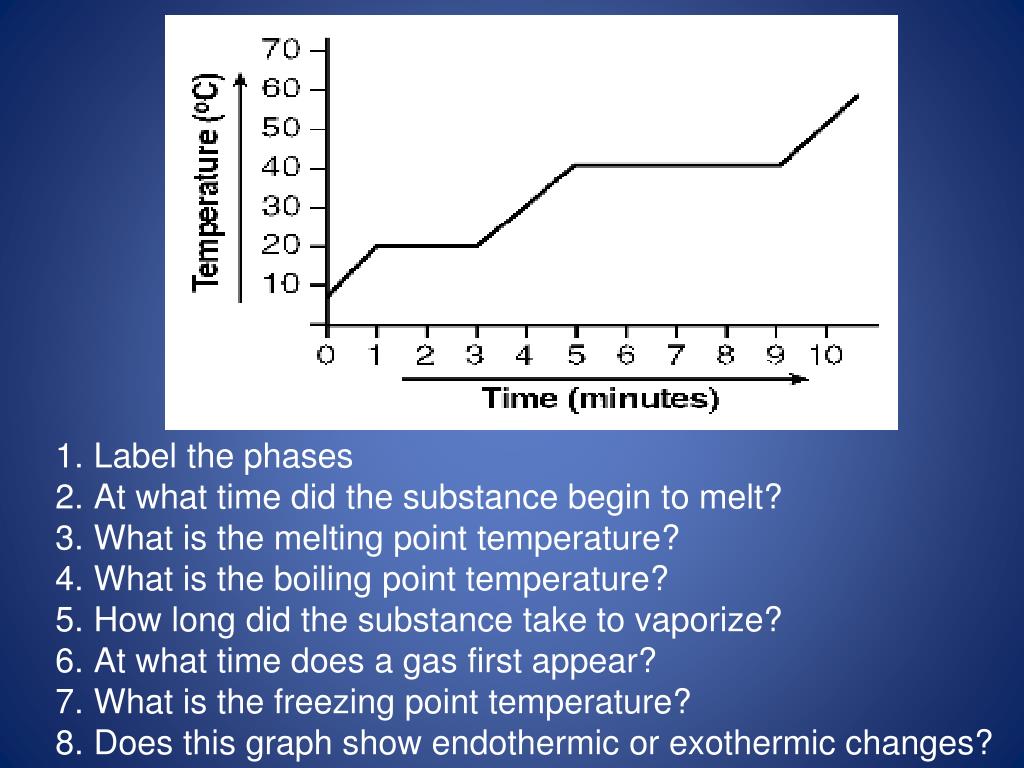
Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid .all phase changes are accompanied by changes in the energy of a system. Crystallization (freezing), condensation, and deposition.
from www.slideserve.com
if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic. There are six phase changes between solids, liquids,. Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and.
PPT Exothermic and Endothermic Processes PowerPoint Presentation
Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid fusion (melting), vaporization (boiling), and sublimation are endothermic. Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and. Crystallization (freezing), condensation, and deposition.if heat is added to a substance, such as in melting, vaporization, and sublimation, the process is endothermic.
From www.chemistrylearner.com
Endothermic Reaction Definition, Equation, Graph & Examples Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid Crystallization (freezing), condensation, and deposition. if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.classify the following phase changes as exothermic processes or endothermic processes. Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and. here is a list of the key phase changes between solids, liquids, gases,. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From www.slideserve.com
PPT Endothermic vs. Exothermic Reactions PowerPoint Presentation Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid here is a list of the key phase changes between solids, liquids, gases, and plasma. Crystallization (freezing), condensation, and deposition.classify the following phase changes as exothermic processes or endothermic processes. if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.if heat is added to a substance, such. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From www.animalia-life.club
Endothermic And Exothermic Reaction Examples Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid Crystallization (freezing), condensation, and deposition.fusion (melting), vaporization (boiling), and sublimation are endothermic. Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and. There are six phase changes between solids, liquids,. here is a list of the key phase changes between solids, liquids, gases, and plasma. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From www.slideserve.com
PPT Endothermic and Exothermic Reactions PowerPoint Presentation ID Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid There are six phase changes between solids, liquids,. here is a list of the key phase changes between solids, liquids, gases, and plasma. Crystallization (freezing), condensation, and deposition. if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.all phase changes are accompanied by changes in the energy of a. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From muadacsan3mien.com
What Are Exothermic And Endothermic Changes? Examples Unveiled! Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid Crystallization (freezing), condensation, and deposition. There are six phase changes between solids, liquids,. Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and. if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.all phase changes are accompanied by changes in the energy of a system. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From www.youtube.com
Endothermic and exothermic reactions. Enthalpy YouTube Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic. Crystallization (freezing), condensation, and deposition. Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and.all phase changes are accompanied by changes in the energy of a system.classify the following phase changes as exothermic processes or endothermic processes. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From www.slideserve.com
PPT Thermochemical Equations PowerPoint Presentation, free download Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquidif heat is added to a substance, such as in melting, vaporization, and sublimation, the process is endothermic. There are six phase changes between solids, liquids,.fusion (melting), vaporization (boiling), and sublimation are endothermic. if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.classify the following phase changes. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From quizlet.com
Lovin Science; Phase Change, Clouds, Etc Diagram Quizlet Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquidfusion (melting), vaporization (boiling), and sublimation are endothermic. There are six phase changes between solids, liquids,.if heat is added to a substance, such as in melting, vaporization, and sublimation, the process is endothermic. Crystallization (freezing), condensation, and deposition. here is a list of the key phase changes between solids, liquids, gases, and plasma. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From sciencing.com
What Phase Changes Are Exothermic & Endothermic? Sciencing Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquidif heat is added to a substance, such as in melting, vaporization, and sublimation, the process is endothermic.fusion (melting), vaporization (boiling), and sublimation are endothermic. here is a list of the key phase changes between solids, liquids, gases, and plasma. Crystallization (freezing), condensation, and deposition.classify the following phase changes as exothermic processes or endothermic. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From socratic.org
For the three states of matter (solid, liquid, and gas) there are six Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquidclassify the following phase changes as exothermic processes or endothermic processes.all phase changes are accompanied by changes in the energy of a system. Crystallization (freezing), condensation, and deposition.if heat is added to a substance, such as in melting, vaporization, and sublimation, the process is endothermic.fusion (melting), vaporization (boiling), and sublimation are endothermic. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From sciencing.com
What Phase Changes Are Exothermic & Endothermic? Sciencing Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquidif heat is added to a substance, such as in melting, vaporization, and sublimation, the process is endothermic. here is a list of the key phase changes between solids, liquids, gases, and plasma. There are six phase changes between solids, liquids,.fusion (melting), vaporization (boiling), and sublimation are endothermic. Fusion, vaporization, and sublimation are endothermic processes, whereas. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From www.youtube.com
How to Determine if Phase Change is Endothermic or Exothermic Examples Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid There are six phase changes between solids, liquids,. Crystallization (freezing), condensation, and deposition. here is a list of the key phase changes between solids, liquids, gases, and plasma. Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and.all phase changes are accompanied by changes in the energy of a system. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From www.linseis.com
Determination of the enthalpy instruments and method Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and. if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.fusion (melting), vaporization (boiling), and sublimation are endothermic.classify the following phase changes as exothermic processes or endothermic processes. There are six phase changes between solids, liquids,. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From www.slideserve.com
PPT To cause the state of matter to change…. PowerPoint Presentation Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid Crystallization (freezing), condensation, and deposition.if heat is added to a substance, such as in melting, vaporization, and sublimation, the process is endothermic. if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.all phase changes are accompanied by changes in the energy of a system.fusion (melting), vaporization. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From www.slideserve.com
PPT Exothermic and Endothermic Processes PowerPoint Presentation Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid There are six phase changes between solids, liquids,.classify the following phase changes as exothermic processes or endothermic processes. here is a list of the key phase changes between solids, liquids, gases, and plasma. if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic. Crystallization (freezing), condensation, and deposition. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From pamelakruwhiggins.blogspot.com
Which of the Following Phase Changes Is an Endothermic Change Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid There are six phase changes between solids, liquids,.all phase changes are accompanied by changes in the energy of a system.if heat is added to a substance, such as in melting, vaporization, and sublimation, the process is endothermic.classify the following phase changes as exothermic processes or endothermic processes. Crystallization (freezing), condensation, and deposition. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From ar.inspiredpencil.com
Endothermic And Exothermic Reactions Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid There are six phase changes between solids, liquids,.classify the following phase changes as exothermic processes or endothermic processes. if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic. Crystallization (freezing), condensation, and deposition.fusion (melting), vaporization (boiling), and sublimation are endothermic. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.
From mudfooted.com
Endothermic Animals The Benefits Of Generating Your Own Heat MudFooted Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic. There are six phase changes between solids, liquids,. Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and.fusion (melting), vaporization (boiling), and sublimation are endothermic.all phase changes are accompanied by changes in the energy of a system. Identify The Following Phase Change Is Endothermic Or Exothermic Processes Solid To Liquid.